Molecular and Reproductive Toxicology
The Research Group for Molecular and Reproductive Toxicology studies how environmental chemicals can disrupt normal development and cause disease. The group has a particular focus on endocrine disruptors and how exposure to these can cause reproductive or cognitive disorders. The ultimate goal is to safeguard human health by improving chemical safety testing and risk assessment.
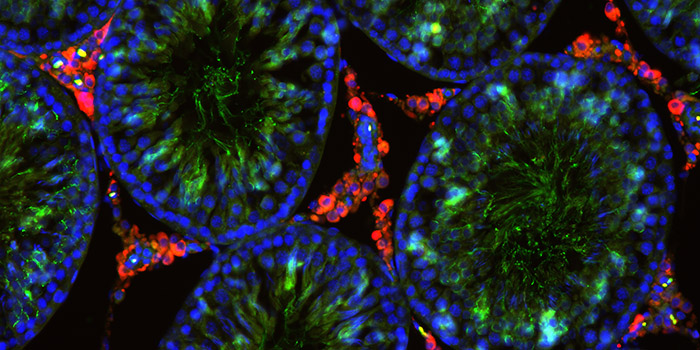
Environmental chemicals with endocrine disrupting properties can be detrimental to normal development and lead to numerous diseases later in life. The Research Group for Molecular and Reproductive Toxicology conducts research aimed at characterizing how such chemicals disrupt development at the molecular level. The group studies the disruption to the sex hormone signaling pathways and reproductive disorders in detail. Disruption of the thyroid hormone system and its consequences for brain development is another focus area.
The research group employs a broad range of methods, from in vitro assays, to organ cultures and rodent in vivo studies. This makes it possible to both define the harmful health effects that can arise from early life exposure to endocrine disruptors and describe how these effects develop. The aim of this work is to elucidate how single chemicals contribute to disease, as well as complex chemical mixtures that are more representative of actual human exposure scenario.
In addition to the basic research portfolio, the group contributes to the improvement of methods for chemical hazard identification and safety evaluations. The group is involved in regulatory toxicology at the international stage and provides expert advice to Danish authorities (The Danish Environmental Protection Agency and the Danish Veterinary and Food Administration).
Contact
Terje Svingen Professor, Head of Research Group Mobile: +45 93518880 tesv@food.dtu.dk